Genetic variants that can act as switches leading to changes in the structure of the RNA molecules that code for proteins have been experimentally validated in plants for the first time. The changes to RNA structure can affect the molecule’s stability, how it interacts with other molecules, and how efficiently it can be translated into protein—all of which can impact its function and the traits of the plant. These genetic switches could be an important genetic mechanism that allowed plants to adapt to their microclimates in the past and could be vital for future adaptation or the development of resilient crops as climates continue to change.
A paper describing the research, led by scientists at Penn State, appears in the journal Genome Biology.
“Proteins, which are one of the main structural and functional molecules of life, are encoded by RNA, which is in turn encoded by DNA,” said Sarah M. Assmann, Waller Professor of Biology at Penn State and a leader of the research team. “Changes in DNA sequence can therefore lead to changes in proteins through an RNA intermediary, but not all DNA changes directly affect the protein. Recently, genetic variants that don’t necessarily change the encoded protein, but instead alter RNA folding, have been associated with human disease. We set out to determine if similar mechanisms existed in plants and if they could be dependent on environmental variables.”
DNA is a double-stranded molecule—it resembles a twisted ladder, with the side rails representing the two strands and the rungs representing bonds that hold them together. RNA, on the other hand, is single-stranded—imagine the ladder cut in half through the middle of the rungs. The single-stranded RNA is generally not just a long, linear molecule, though. It folds back on itself forming short double-stranded sections between loops and bubbles of single-stranded RNA. This secondary folded structure is determined by the RNA sequence in conjunction with the cellular microenvironment and is important to RNA function.
The folded structure of an RNA molecule can therefore be altered by genetic variants known as “single-nucleotide polymorphisms” or SNPs—places in the genome where a single letter in the DNA alphabet differs between two or more individuals or groups. These RNA-structure-altering SNPs are known as “riboSNitches,” combining “ribo” from the R in RNA, “SNP”, and “switches.”
“We study Arabidopsis, a model organism for plant biology,” said Ángel Ferrero‑Serrano, assistant research professor of biology at Penn State and first author of the paper. “Over the last decade, the advent of high-throughput technologies is generating vast amounts of genetic data and physical descriptions of Arabidopsis varieties collected across the native range of the species. Specimens of Arabidopsis from locations across the globe have had their whole genomes sequenced. We recently developed a set of computational tools, CLIMtools, that allows us to determine associations between the genetic variation in DNA across Arabidopsis varieties collected within their native range and a large set of climate variables that define the local environments of those varieties. We used these tools to find SNPs associated with temperature variables and then tested whether any of the SNPs acted as riboSNitches.”
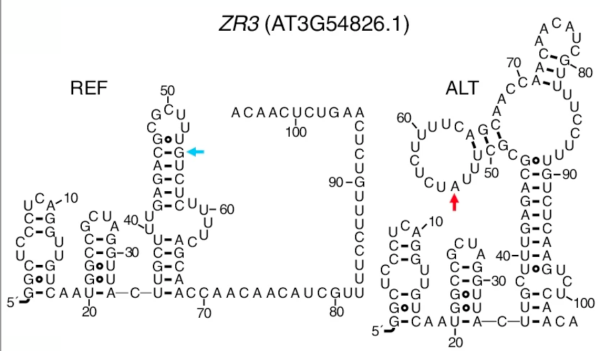
The team took the set of SNPs associated with changes in temperature and further narrowed it by looking for SNPs that were also associated with changes in RNA abundance, which often result from changes in RNA folding. They then applied an RNA structure algorithm to see if any of the SNPs were predicted to lead to structural changes, and chose two genes to experimentally validate.
“We tested the stability of short synthetic RNA molecules that include the potential riboSNitches as compared to the standard, reference sequence of that same stretch of RNA, across a range of temperatures,” said Ferrero-Serrano. “Based on our experiments, the SNPs in both of the genes appear to act as riboSNitches, one of which in particular alters the stability of the RNA structure in a temperature-dependent fashion. We suggest the term ‘conditional riboSNitches’ to denote riboSNitches that are dependent on environmental variables.”
Having experimentally demonstrated the existence of riboSNitches in Arabidopsis, the team then performed a massive computational study to predict potential riboSNitches across the genomes of hundreds of different sequenced Arabidopsis varieties. Of over 3.8 million SNPs that were assessed, over a million, or about 27 percent, have the potential to act as riboSNitches.
“Plants can’t move around, so adaptation to their local environments promotes survival,” said Assmann. “We now know that riboSNitches are another arrow in the quiver of genetic tools available to plants for that adaptation and that they can be conditional on environmental variables.”
With CLIMtools, the team have created a set of resources to study the relationships between genetic variation in Arabidopsis and the environment. They hope this will allow the scientific community to better understand how plants have adapted to local environments and how they can continue to thrive as climates change.
“Easy access to data is going to be critical to solving current challenges to agriculture in a sustainable way,” said Dr. Doreen Ware, a USDA ARS scientist at Cold Spring Harbor Laboratory. “We are excited to be hosting CLIMtools within Gramene, our online portal for comparative functional genomics, as it contains new resources to access information on standing genetic variation that will be useful for climate adaptation, as well as information that can be used for gene editing approaches to create new alleles in agriculturally important crops.”
The techniques used for this study range from biophysics to molecular biology to ecology.
“We brought together a diverse team of researchers at Penn State who ranged from an undergraduate, to a graduate student, to a research professor, and it was a pleasure to work with them and Professor Assmann as well as our colleagues at Cold Spring Harbor Laboratory,” said Philip C. Bevilacqua, Distinguished Professor of Chemistry and of Biochemistry and Molecular Biology at Penn State. “The findings that variation in sequence can be manifested in variation in RNA structure, has long term implications for modulation of crop yield in the face of adverse climate conditions.”
In addition to Assmann, Ferrero‑Serrano, Ware, and Bevilacqua, the research team includes plant biology graduate student Megan M. Sylvia and biochemistry and molecular biology undergraduate Peter C. Forstmeier at Penn State; and principal engineer Andrew J. Olson at Cold Spring Harbor Laboratory in NY. The research was funded by Penn State, the U.S. National Science Foundation and the U.S. Department of Agriculture.
